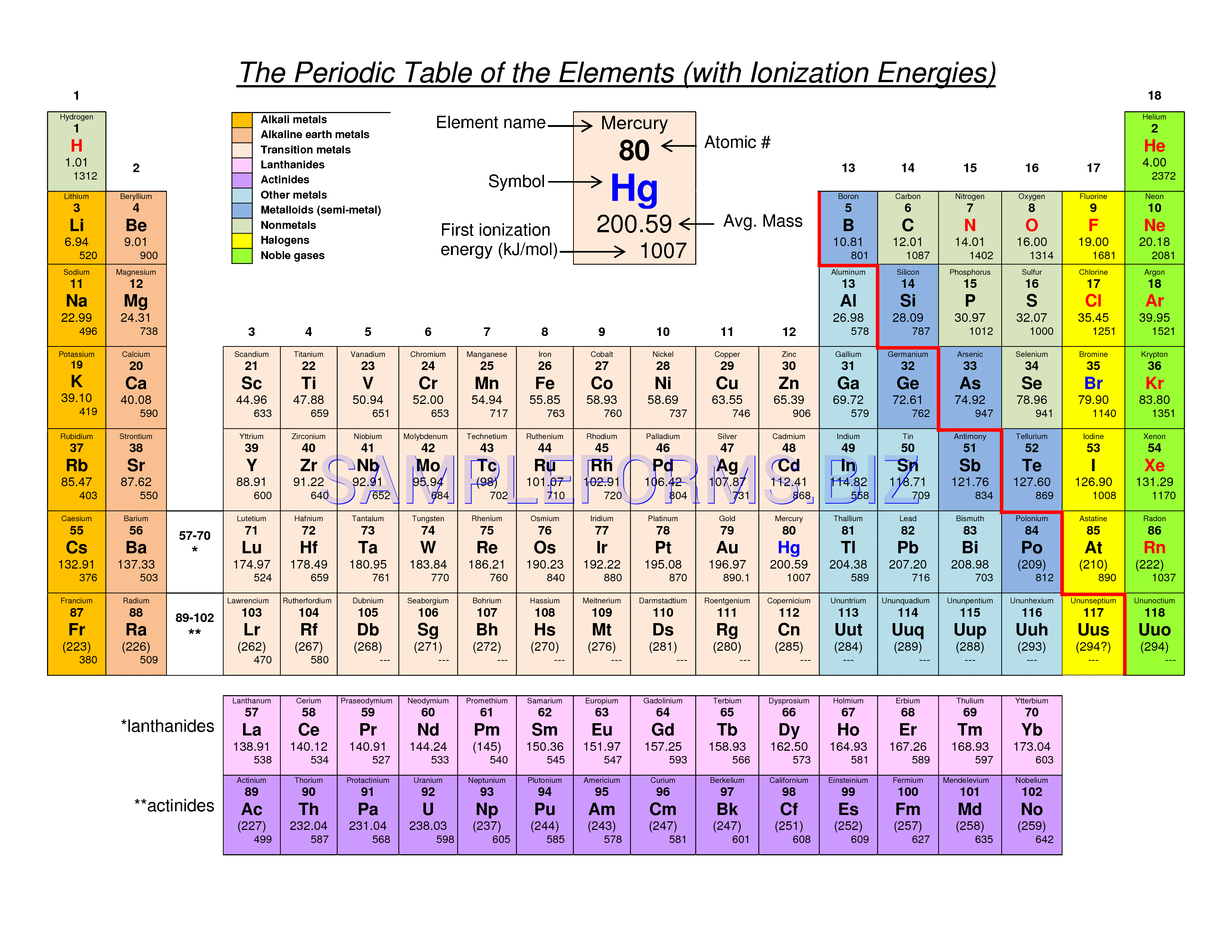
In every ionic compound, the total number of positive charges of the cations equals the total number of negative charges of the anions. For example, copper can form ions with a 1+ or 2+ charge, and iron can form ions with a 2+ or 3+ charge. Transition metals and some other metals often exhibit variable charges that are not predictable by their location in the table. The name of the anions is the name of the nonmetallic element with its ending replaced by the suffix -ide, so O 2− is called oxide. The anion, O 2−, has the same number of electrons as the next noble gas – neon. 
This results in an anion with 8 protons, 10 electrons, and a 2− charge, and is symbolized as O 2−. 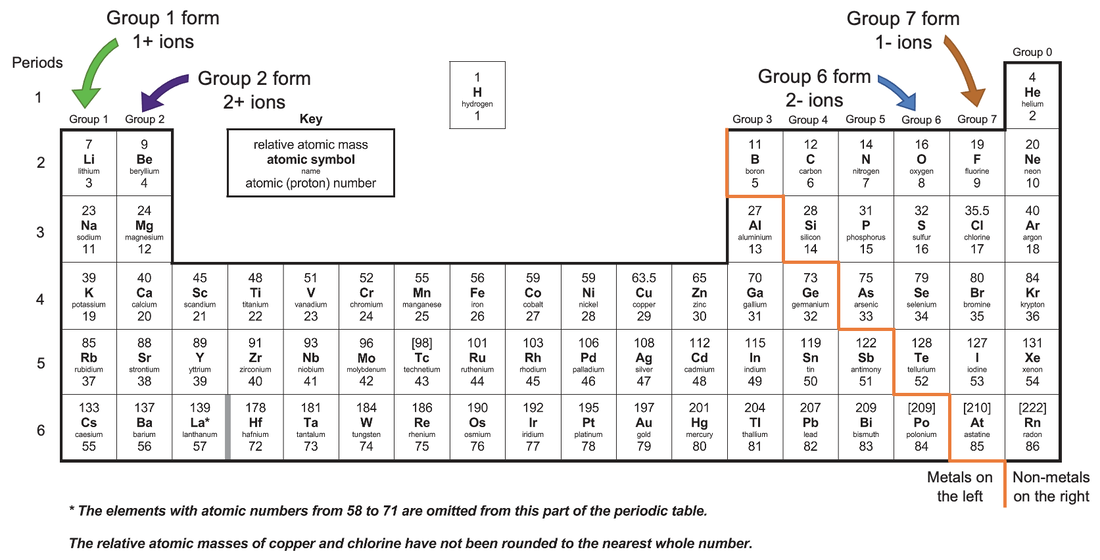
For example, a neutral oxygen atom, with 8 protons and 8 electrons, readily gains two electrons. That is, atoms of group 17 gain one electron and form anions with a 1− charge atoms of group 16 gain two electrons and form ions with a 2− charge, and so on. Moving from the far right to the left on the periodic table, nonmetal elements often gain electrons to form anions with the same number of electrons as an atom of the next noble gas in the periodic table, and a negative charge equal to the number of groups moved left from the noble gases. The compound formed by this transfer is stabilized by the electrostatic attractions (ionic bonds) between the oppositely charged ions. An element composed of atoms that readily lose electrons (a metal) can react with an element composed of atoms that readily gain electrons (a nonmetal) to produce ions through complete electron transfer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
